23 May 2013
23 May 2013
 J. Agric. Food Chem.61 - 26, pp. 6533 - 6543.
J. Agric. Food Chem.61 - 26, pp. 6533 - 6543.
 Sonia Losada-Barreirob, Fátima Paiva-Martinsa, Carlos Bravo-Díaz*b, Laurence S. Romstedc
Sonia Losada-Barreirob, Fátima Paiva-Martinsa, Carlos Bravo-Díaz*b, Laurence S. Romstedc
 Autor affilations:
Autor affilations:
*Corresponding authors
aREQUIMTE-LAQV, Departamento de Química e Bioquímica, Faculdade de Ciências, Universidade do Porto, 4169-007, Portugal
bDpt. Química Física, Facultad de Química, Universidad de Vigo, Vigo-Pontevedra, Spain
cDepartment of Chemistry and Chemical Biology, Rutgers, The State University of New Jersey, Piscataway, New Jersey 08854, United States
![]() E-mail: cbravo@uvigo.es
E-mail: cbravo@uvigo.es
Abstract
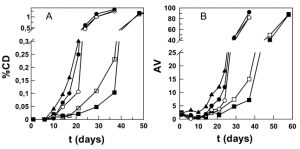
Antioxidant (AO) efficiencies are reported to go through maxima with increasing chain length (hydrophobicity) in emulsions. The so-called “cutoff” after the maxima, indicating a decrease in efficiency, remains unexplained. This paper shows, for gallic acid (GA) and propyl, octyl, and lauryl gallates (PG, OG, and LG, respectively), that at any given volume fraction of emulsifier, the concentrations of antioxidants in the interfacial region of stripped corn oil emulsions and their efficiency order follow PG > GA > OG > LG. These results provide clear evidence that an AO’s efficiency correlates with its fraction in the interfacial region. AO distributions were obtained in intact emulsions by using the pseudophase kinetic model to interpret changes in observed rate constants of the AOs with a chemical probe, and their efficiencies were measured by employing the Schaal oven test. The model provides a natural explanation for the maxima with increasing AO hydrophobicity.